|
Do not proceed to schedule a custom demo unless you have already conferred with the lecture demonstrator about it. 
The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. So, the reactivity series of metals can be defined as a series of metals, in order of reactivity from highest to lowest. The university shall not be liable for any special, direct, indirect, incidental, or consequential damages of any kind whatsoever (including, without limitation, attorney's fees) in any way due to, resulting from, or arising in connection with the use of or inability to use the web site or the content. Reactivity series is the series of metals based on their reactivity from highest to lowest. The university further disclaims all responsibility for any loss, injury, claim, liability, or damage of any kind resulting from, arising out or or any way related to (a) any errors in or omissions from this web site and the content, including but not limited to technical inaccuracies and typographical errors, or (b) your use of this web site and the information contained in this web site.the university shall not be liable for any loss, injury, claim, liability, or damage of any kind resulting from your use of the web site. 
The university expressly disclaims all warranties, including the warranties of merchantability, fitness for a particular purpose and non-infringement. So, if one metal is going to lose its electrons, another species (a metal ion) has to pick them up. This web site is provided on an "as is" basis. Metals (in their elemental form) don’t react with each other. Certain elements react more easily with some elements than others. Activity Series: An easy way to use the table is to note that a metal can reduce any ion of a metal below it in the table. "Do not do demos unless you are an experienced chemist!" Updated: Single Displacement Reactions In chemistry, elements react to form products. The ranking of metals (and hydrogen) based on their activity is called the activity series. In summary, the electronegativity of an atom affects the reactivity whether it needs to gain or lose electrons to react.Please read the following disclaimer carefullyīy continuing to view the descriptions of the demonstrations you have agreed to the following disclaimer. This would mean that a higher electronegativity would give it a higher ability to gain electrons and hence react more readily, placing them higher in the reactivity series, unlike the metallic elements that would go through a different scenario. 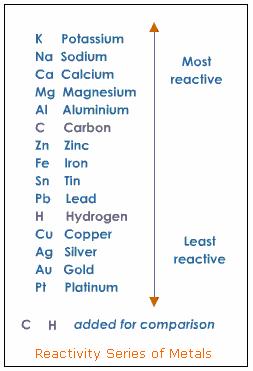
The reason why it is different for the non metallic elements is that they need to gain electrons in order to react and form bonds. Virtual Lab: Activity Series of Metals Name: Write and balance the formula equation for the following reaction: Zinc metal reacts with a lead(II) nitrate solution to form a zinc nitrate solution and a lead precipitate. The magnesium will lose the electrons less readily and so it will be placed lower than sodium in the reactivity series. The magnesium ion has charge of 2+ while Na has a charge of 1+, this would mean that the there is stronger attraction between the valence electrons of Mg than the valence electrons of Na. (For this you need to consider only the net charge of the ions formed as the charge would be considered neutral once electrons and protons are equal). There are also other cases in the reactivity series that are explained in a similar way. The reaction would tend to occur more readily hence the reactivity of potassium is higher than sodium. The potassium would have a lower tendency of attracting electrons compared to sodium, it would hence more readily lose electrons to form potassium ions (K+) to form ionic bonds with other elements. 
Top-ranked metals displace the ions of metals lower in the series from solution. However, as you said the reactivity series says potassium is more reactive than sodium. The series ranks metals by oxidation potential. Here is a visual explanation of the situation.Īs the atoms move from left to right, the electronegativity of these atoms are decreasing as the distance between the electrons and the positively charged nucleus is would be greater, giving it lower ability to attract electrons. Whereas an element's reactivity is based more on how ready it is to gain or lose electrons. Reactivity series In chemistry, a reactivity series (or activity series) is an empirical, calculated, and structurally analytical progression 1 of a series of metals, arranged by their 'reactivity' from highest to lowest. 1 An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. Electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
